- HOME
- ABOUT
- AREA OF FOCUS
- Research
- Enteric diseases & HIV vaccine Research unit
- HIV prevention, care & treatment
- Implementation Science
- Paediatric prevention care & treatment
- Vulnerable Children & Adolescent Health
- Tuberculosis
- Prison’s Health
- Social & Behavioral studies
- New Born & Women’s health
- Primary care and health systems strengthening
- Mental Health
- Hepatitis
- SUPPORT UNITS
- MEDIA
- News
- Annual reports
- Quarterly Newsletters
- Vol.24 CIDRZ Newsletter Q2 FY 2024
- Vol.23 CIDRZ Newsletter Q1 FY 2024
- Vol.22 CIDRZ Newsletter Q4 FY 2023
- Vol.21 CIDRZ Newsletter Q3 FY 2023
- Vol.20 CIDRZ Newsletter Q2 FY 2023
- 1st Quarter Newsletter FY2022/2023
- 4th Quarter Newsletter FY2021/2022
- 3rd Quarter Newsletter FY 2021/2022
- 2nd Quarter – FY 2021-2022
- 1st Quarter 2021-2022
- Research Abstracts/ Scientific Posters
- Research Presentations
- Gallery
- Videos
- TRAINING
- TOOLKITS
- Taskpen Toolkit
- Human Infection Studies (HIS)
- Menstrual Hygiene Management (MHM)
- Better Information for Health in Zambia Toolkit – 2017
- CommART Toolkit
- Operation Triple Zero Plus – HIV Literacy Package for Adolescents
- TASKPEN CLINICAL GUIDELINES :Protocols for the Integrated Management of Cardiometabolic conditions in Adult PLHIV
- JOBS
Uncategorized
- Filter by
- Categories
- Tags
- Authors
- Show all
- All
- Announcements
- Blog
- CIDRZ Central Laboratory
- CIDRZ FrontPage
- Frontpage Article
- In The Press
- Latest News
- News
- Presentations
- Programs
- Publications
- Research Rounds
- Success Stories
- TB NATIONAL INTER-SCHOOLS SINGING COMPETITION
- Uncategorized
- All
- 10 years of VMMC
- 90:90:90 Targets
- Academy Health
- Accreditaion
- ACHIEVE
- Adolescent HIV in Zambia
- AIDS
- Airtel
- Airtel Zambia
- Akazi
- Apoorva Mehrotra
- Atlas Mara Bank
- BetterInfo study
- Bill and Melinda Gates foundation
- Birth Certificates
- Bongo Hive
- C-CoHP Project
- CDC
- Central Laboratory
- Centre for Infectious Disease Research in Zambia
- Cervical Cancer
- CETA
- Chainda Primary School
- CHEKUP I Project
- Chongwe District hospital
- CIDRZ
- CIDRZ LAB
- CIDRZ Research
- Circumcision
- Clinical Research Site
- CMMB
- collaboration
- Common Elements Treatment Approach
- Community Comprehensive Health Package for Adolescents and Young People
- communityoutreach
- COVID-19
- Covid-19 testing
- COVID-19 Vaccine
- COVID19
- COVPN
- CQUIN’s 6th Annual Meeting
- CSR
- CTL specificities and HLA class I allele
- Dr Anthony Fauci
- Dr Chabuka Jerome Kawesha
- Dr Izukanji Sikazwe
- Dr. Gene Bukhman
- DREAMS
- DSD
- ECAP III
- ECAPIII
- EGC machines
- equality
- Estee Launder Group of Companies
- evidence-based treatments
- Expanded Programmes on Immunisations
- Gavi
- Girls 4 Girls (G4G)
- Global Alliance for Vaccines and Immunisation (Gavi)
- Global Fund
- Havard University
- Health Care Workers
- HIV
- HIV Prevention treatment and care
- HIV unmuted
- HIV/AIDS Prevention in Zambia
- HIV/AIDS Program in Zambia
- HVTN
- Immunisation
- Implementation Science
- IS Conference 2023
- James Koni
- Key Populations
- Lab
- Live webinar
- Livingstone District Health Office
- LOGISTIMO
- LPHO
- LUSAKA DISTRICT EDUCATION BOARD
- Lusaka Provincial Health Office
- MAC cosmetics
- MCDSS
- Men's Clinics
- Men's Healt
- mental disorders
- Mental health
- Ministry of Community Development and Social Services
- Ministry of Community Development and Social Services (MCDSS)
- Ministry of Education
- Ministry of Health
- MOH
- MoH Child Health Unit
- NAC
- National Institute of Health (NIH)
- NCD
- NCDI
- NHIMA
- Northwestern Province
- NRC
- Omicron
- Oxygen
- Oxygen cylinders
- P2G
- Pact Inc
- Paediatric Care and Treatment
- Patient Centred Care
- PCC
- PCZ
- PEN Plus
- PEN plus project
- PEPFAR
- Person Centred Care
- Pfizer Vaccine
- Pharmacy
- PMTC
- Pregnant Parenting Girls
- Psychological First Aid
- publichealth
- Research studies
- Sexual Reproductive Health Services and Rights
- ShangRing
- soccer Gala
- Southern Province
- SRHSR
- Strategic Information
- TB
- TB Champion
- TBLON
- TBLON Project
- TDRC
- Therapeutic Art
- Tropical Diseases Research Centre
- Tuberculosis Local Organisations Network
- UNAIDS
- UNFPA
- UNICEF
- USAID
- USAID CHEKUP I Project
- USAID Zambia
- Vaccine
- Vaccine awareness
- Vaccines
- Vaccines Against Shigella & ETEC (VASE) Conference
- VASE
- ViiV Health Care
- VMMC
- vulnerablepopulations
- Western Province
- WHO
- World AIDS Day
- Young Women Christian Association
- YWCA
- ZACCI
- Zambia Correctional Service
- Zambia Medical Association
- Zambia Police
- Zambia Private Sector Alliance
- ZCS
- ZMA
- ZNPHI
- ZTA
September 12, 2023
September 12, 2023
Infection Prevention and Control (IPC) is critical in health facilities to avoid transmission of diseases. However, IPC measures cannot be well executed without competently trained healthcare […]
July 27, 2023
July 27, 2023
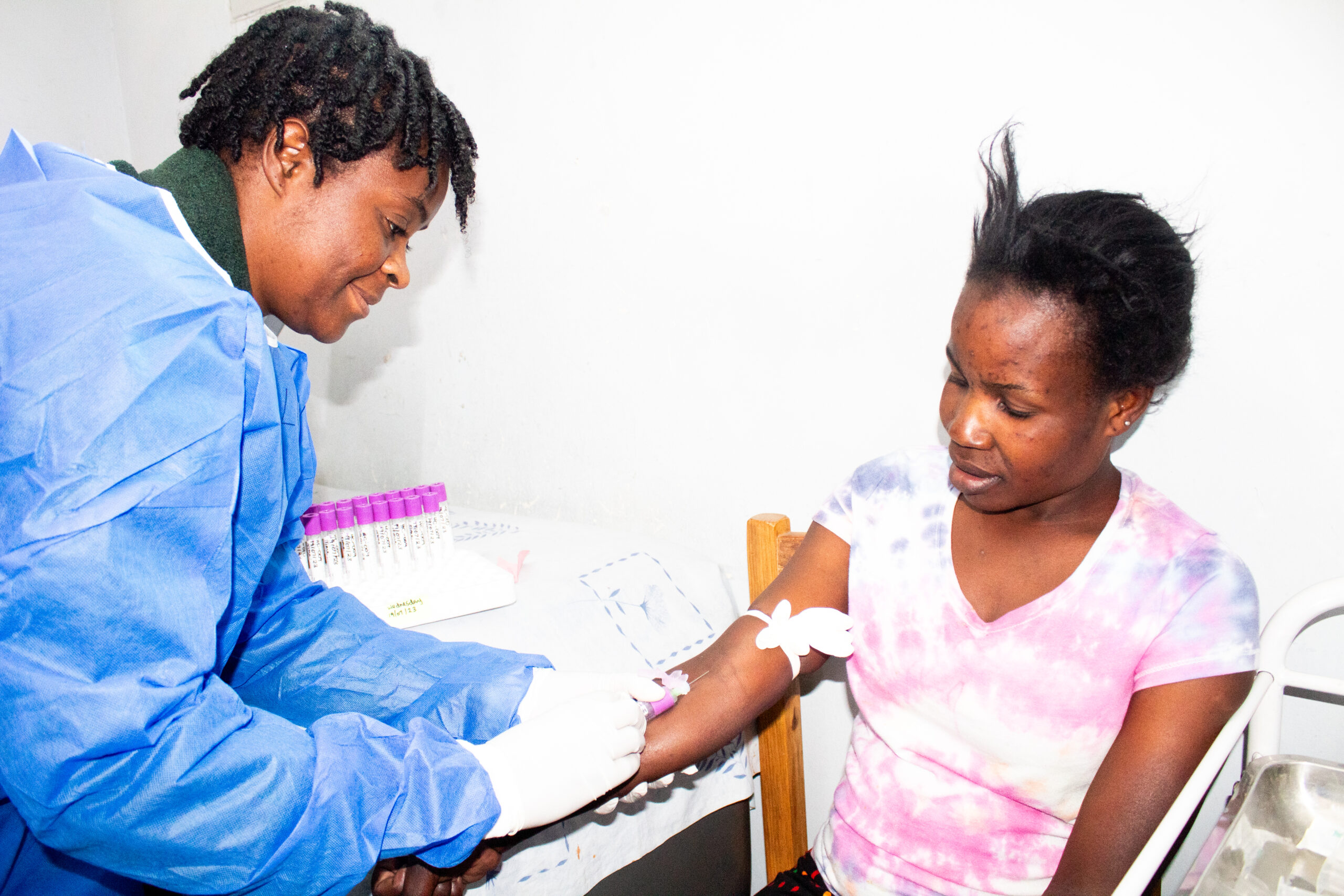
The CIDRZ research team has joined other researchers from Kenya, Rwanda, South Africa and Zimbabwe, in conducting a three-year study which shall help tailor an HIV […]
July 27, 2023
July 27, 2023
The Centre for Infectious Disease Research in Zambia (CIDRZ) hosted visitors from the United Nations Population Fund (UNFPA), the Joint United Nations Programme on HIV/AIDS (UNAIDS), […]
July 17, 2023
July 17, 2023
The media in Zambia have been encouraged to advocate for enhanced Tuberculosis (TB) detection and treatment for the country to meet the global target of ending […]
June 27, 2023
June 27, 2023
Aligned with our mission of improving access to quality healthcare in Zambia through innovative capacity development, the Centre for Infectious Disease Research in Zambia (CIDRZ), through […]
June 22, 2023
June 22, 2023
In a quest to alleviate the devastating effects of mental health in the communities, the Zambia National Public Health Institute (ZNPHI’s) Zambia COVID-19 Emergency Response and […]