CIDRZ Engages Chimwemwe Level 1 Hospital on Diagnostic Service Collaboration
June 24, 2025
CIDRZ Board Chair Applauds Dr Sikazwe’s Transformative Leadership.
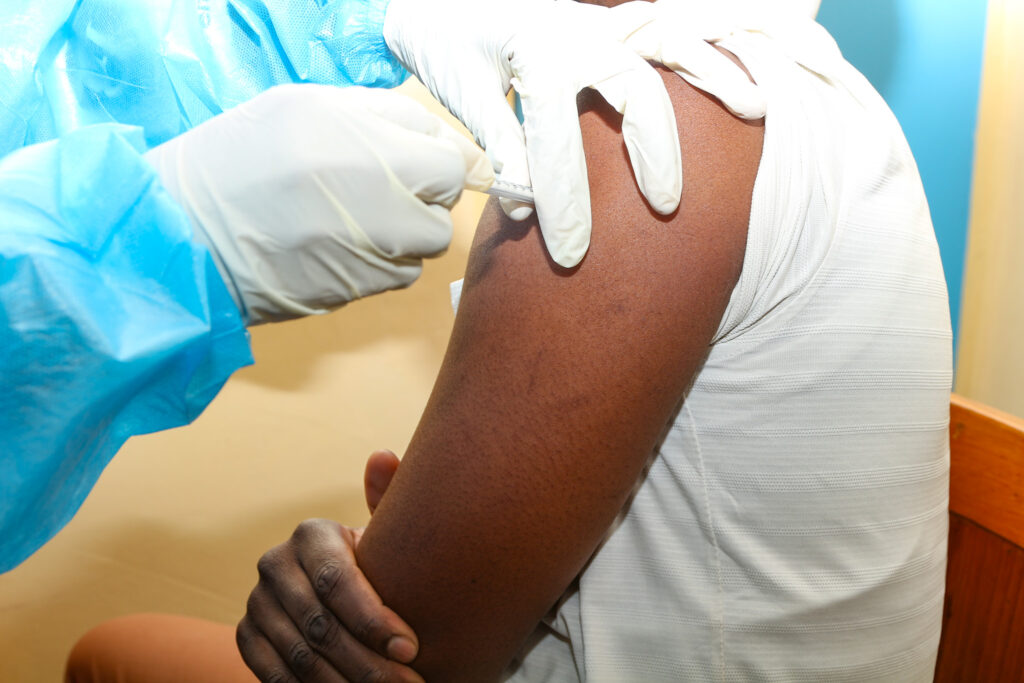
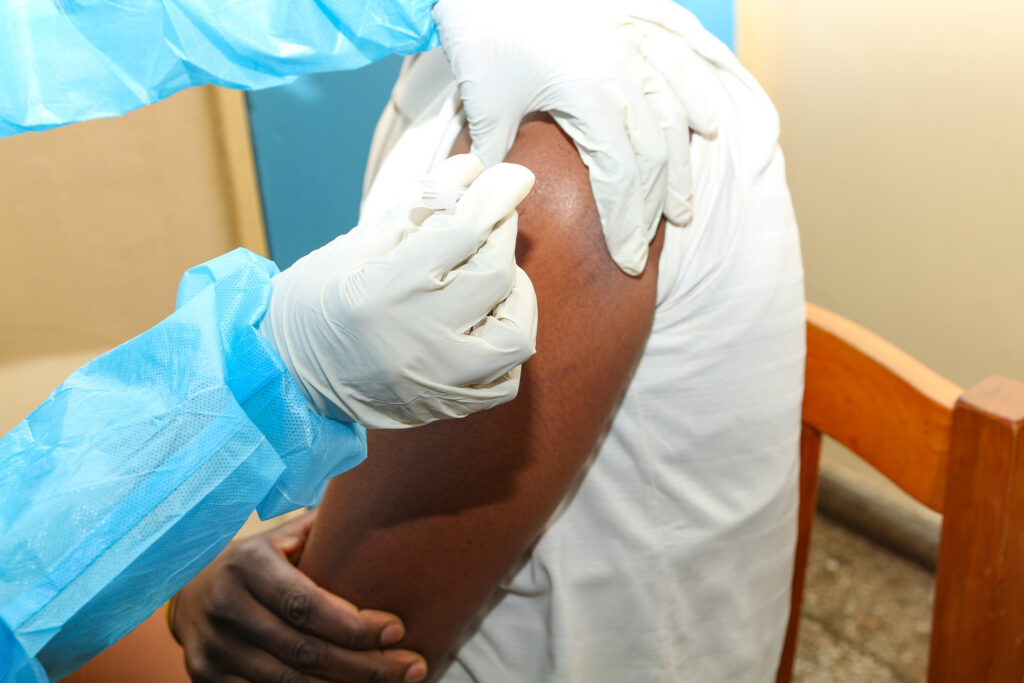
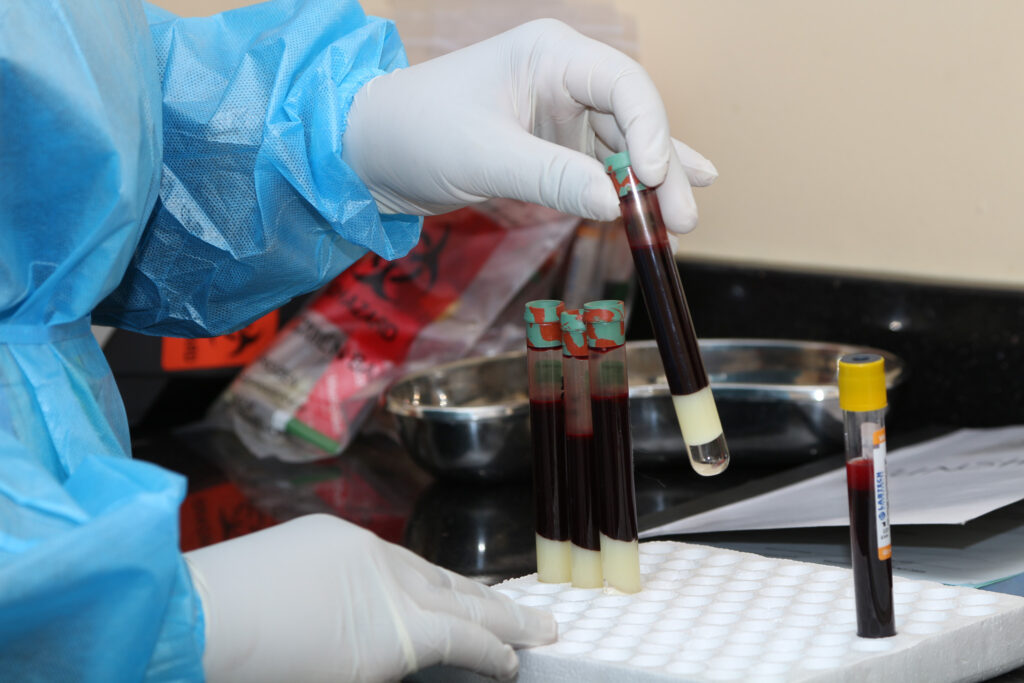
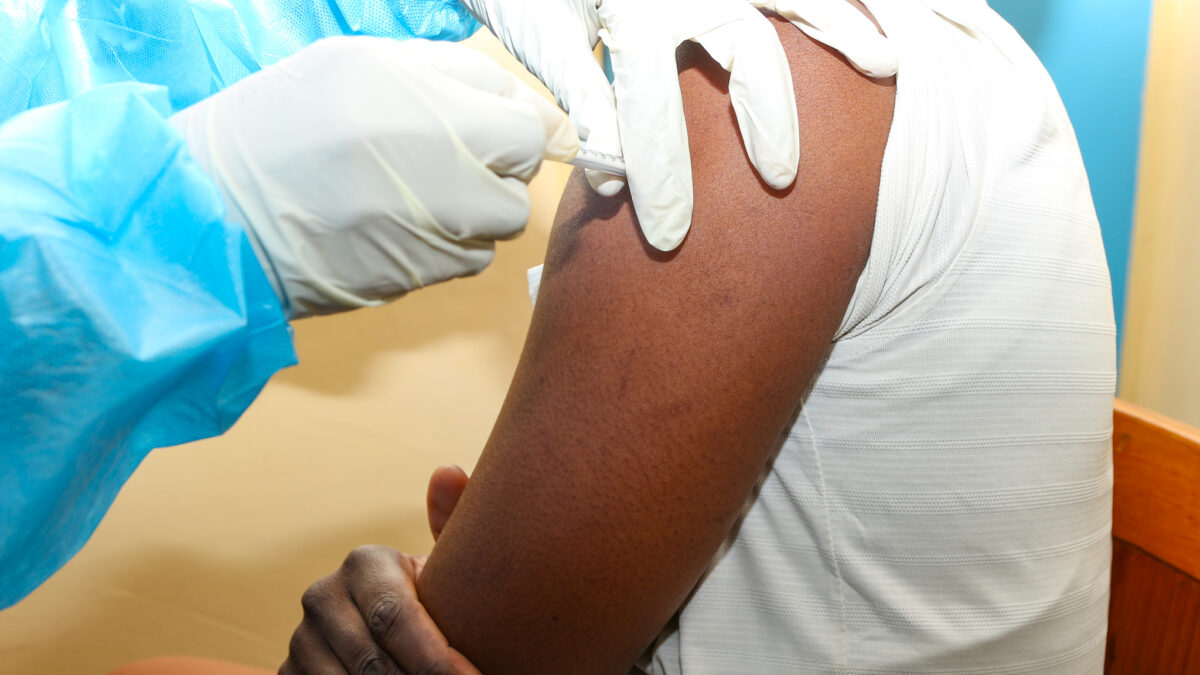
June 26, 2025The Centre for Infectious Disease Research in Zambia (CIDRZ), in collaboration with the Ministry of Health and the the ShigaPlexIM consortium led by European Vaccine Initiative, has successfully administered the first dose of the SUNSHINE Phase 1b clinical trial. The trial is testing the InvaplexAR-Detox/dmLT vaccine, a promising candidate against shigellosis, a severe diarrhoeal disease. The trial, sponsored by Leiden University Medical Centre (LUMC, the Netherlands) who conducted the Phase 1a study, where the safety and dose finding of the vaccine was confirmed, allowing the start of this Phase Ib trial at CIDRZ in Zambia.
This important milestone was achieved at the CIDRZ-run Matero Clinical Research Site in Lusaka, where the trial aims to enrol up to 35 healthy adult volunteers.
The study evaluates the vaccine’s safety and assesses immune response markers over a 28-day follow-up period, with immunological assay support provided by the University of Gothenburg.
Led by Principal Investigator Mr Michelo Simuyandi, the trial is the result of close coordination among a dedicated multidisciplinary team comprising clinical researchers, laboratory scientists, pharmacists, community engagement officers, and data and research nurses.
A successful outcome in this early-stage trial would mark a significant step toward developing a safe, effective, and accessible vaccine for shigellosis, a disease that disproportionately affects children and vulnerable populations in low-resource settings.
Shigellosis, caused by Shigella species, is a leading cause of severe diarrhoea worldwide, with an estimated 80–165 million cases and up to 64,000 deaths annually among children under five in lower-income countries.
Rising antimicrobial resistance has rendered many current treatments less effective, prompting the World Health Organization to prioritise the development of a Shigella vaccine.
Funding: This project is part of the EDCTP2 Programme supported by the European Union (RIA2018V-2308).